|
is a 9-month-old male infant of Ashkenazi Jewish parentage. For prenatal screening, molecular techniques are the preferred method because of their sensitivity and the limited amount of sample available (see Chapter 17 ). This is accomplished by differential heat inactivation of the sample (exposure of the sample to 50☌ will inactivate hexosaminidase A activity but not hexosaminidase B activity). When measuring enzyme activity, one needs to be careful to distinguish between hexosaminidase A activity and a closely related activity owing to hexosaminidase B. , can be assayed using a serum sample and a substrate that releases a fluorescent dye upon being hydrolyzed. Hexosaminidase A, the enzyme activity that is lacking in Jay S. The hexosaminidase A gene encodes the α-chain for the hexosaminidase A protein. 
has an insertion in exon 11 of the α-chain of the hexosaminidase A gene, the most common mutation found in patients of Ashkenazi Jewish background who have Tay–Sachs disease. The results of tests performed in the molecular biology laboratory show that Jay S. Her hemoglobin level was 7.3 g/dL (reference range for females, 12 to 16 g/dL). , a 4-year-old patient with β + -thalassemia intermedia (see Chapter 13 ), showed no improvement in her symptoms at her second visit. These proteins are transferred to the Golgi complex, where they are modified and targeted to their ultimate locations. Proteins synthesized on ribosomes attached to the rough endoplasmic reticulum (RER) are destined for lysosomes, cell membranes, or secretion from the cell. Proteins synthesized on cytosolic ribosomes are released into the cytosol or transported into mitochondria, peroxisomes, and the nucleus. Modification of amino acid residues in a protein occurs during or after translation. This process involves proteins called chaperones. Folding of the polypeptide into its three-dimensional configuration occurs as the polypeptide is being translated. įolding and Modification and Targeting of the Protein. The complex of a single mRNA with multiple ribosomes is known as a polysome. Release factors bind instead, causing the completed protein to be released from the ribosome.Īfter one ribosome binds and moves along the mRNA, translating the polypeptide, another ribosome can bind and begin translation. These three elongation steps are repeated until a termination codon aligns with the site on the ribosome where the next aminoacyl-tRNA would normally bind. It requires guanosine triphosphate ( GTP ) and proteins known as eukaryotic initiation factors (eIFs).Įlongation of the polypeptide involves three steps : (1) binding of an aminoacyl-tRNA to the “A” site on the ribosome, where it base-pairs with the second codon on the mRNA (2) formation of a peptide bond between the first and second amino acids and (3) translocation, movement of the mRNA relative to the ribosome, so that the third mRNA codon moves into the “A” site. Initiation involves the formation of a complex containing the initial methionyl-tRNA bound to the AUG “start” codon of the mRNA and the “P” site of the ribosome. Base pairing between the anticodon of the tRNA and the codon on the mRNA ensures that each amino acid is inserted into the growing polypeptide at the appropriate position. Note that the codon–anticodon pairing is complementary and antiparallel.Įach amino acid is carried to the ribosome by an aminoacyl-transfer RNA ( tRNA ) (i.e., a tRNA with an amino acid covalently attached). The tRNA contains an amino acid at its 3′-end that corresponds to the codon on mRNA with which the anticodon of the tRNA can base-pair. Binding of transfer RNA (tRNA) to a codon on messenger RNA (mRNA). The protein is synthesized from its N -terminus to its C -terminus. The codons on mRNA are read sequentially in the 5′-to-3′ direction, starting with the 5 ′ -AUG (or “ start ” codon) that specifies methionine and sets the reading frame and ending with a 3 ′ -termination (or “ stop ”) codon ( UAG, UGA, or UAA ). The portion of mRNA that specifies the amino acid sequence of the protein is read in codons, which are sets of three nucleotides that specify individual amino acids ( Fig. 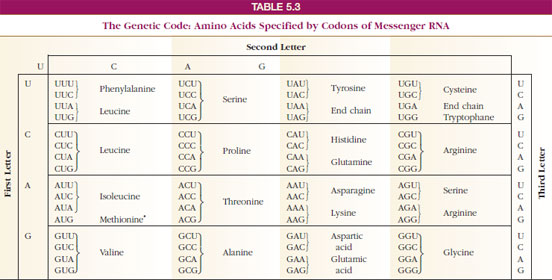  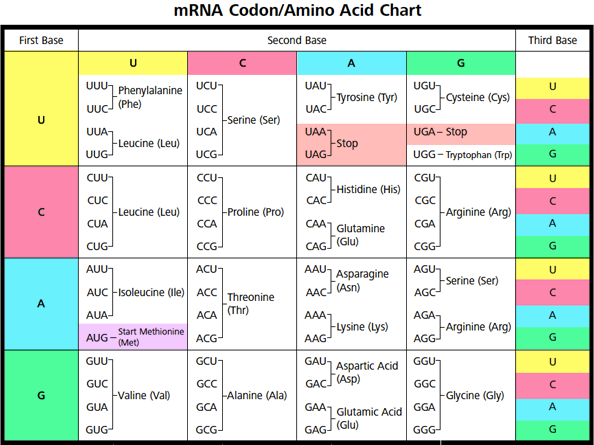
The genetic message encoded in DNA is first transcribed into mRNA, and the nucleotide sequence in the coding region of the mRNA is then translated into the amino acid sequence of the protein. Proteins are produced by the process of translation, which occurs on ribosomes and is directed by messenger RNA ( mRNA ).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
